
This is a large amount of energy for a nuclear reaction the amount is so high because the binding energy per nucleon of the helium-4 nucleus is unusually high because the He-4 nucleus is " doubly magic". Hence, the energy released is 0.0238 × 931 MeV = 22.2 MeV.Įxpressed differently: the mass is reduced by 0.3%, corresponding to 0.3% of 90 PJ/kg is 270 TJ/kg. We first need the energy equivalent of one atomic mass unit: Using Einstein's mass-energy equivalence formula E = mc 2, the amount of energy released can be determined. The "missing" rest mass must therefore reappear as kinetic energy released in the reaction its source is the nuclear binding energy. In a nuclear reaction, the total (relativistic) energy is conserved. the total rest mass on the two helium-nuclei = 2 × 4.0026 = 8.0052 u.the sum of the rest mass of the individual nuclei = 6.015 + 2.014 = 8.029 u.Nucleus has a standard atomic weight of 6.015 atomic mass units (abbreviated u), the deuterium has 2.014 u, and the helium-4 nucleus has 4.0026 u. This can be calculated by reference to a table of very accurate particle rest masses, as follows: according to the reference tables, the 6 Kinetic energy may be released during the course of a reaction ( exothermic reaction) or kinetic energy may have to be supplied for the reaction to take place ( endothermic reaction). The reaction above would be written as 6Li(d,α)α. Common light particles are often abbreviated in this shorthand, typically p for proton, n for neutron, d for deuteron, α representing an alpha particle or helium-4, β for beta particle or electron, γ for gamma photon, etc. This style of the form A(b,c)D is equivalent to A + b producing c + D. Instead of using the full equations in the style above, in many situations a compact notation is used to describe nuclear reactions. Nuclear reactions may be shown in a form similar to chemical equations, for which invariant mass must balance for each side of the equation, and in which transformations of particles must follow certain conservation laws, such as conservation of charge and baryon number (total atomic mass number). The feat was popularly known as "splitting the atom", although it was not the modern nuclear fission reaction later (in 1938) discovered in heavy elements by the German scientists Otto Hahn, Lise Meitner, and Fritz Strassmann. Eventually, in 1932 at Cambridge University, a fully artificial nuclear reaction and nuclear transmutation was achieved by Rutherford's colleagues John Cockcroft and Ernest Walton, who used artificially accelerated protons against lithium-7, to split the nucleus into two alpha particles. This was the first observation of an induced nuclear reaction, that is, a reaction in which particles from one decay are used to transform another atomic nucleus. In 1919, Ernest Rutherford was able to accomplish transmutation of nitrogen into oxygen at the University of Manchester, using alpha particles directed at nitrogen 14N + α → 17O + p. Various nuclear fusion reactions of light elements power the energy production of the Sun and stars. Nuclear chain reactions in fissionable materials produce induced nuclear fission. Natural nuclear reactions occur in the interaction between cosmic rays and matter, and nuclear reactions can be employed artificially to obtain nuclear energy, at an adjustable rate, on-demand.  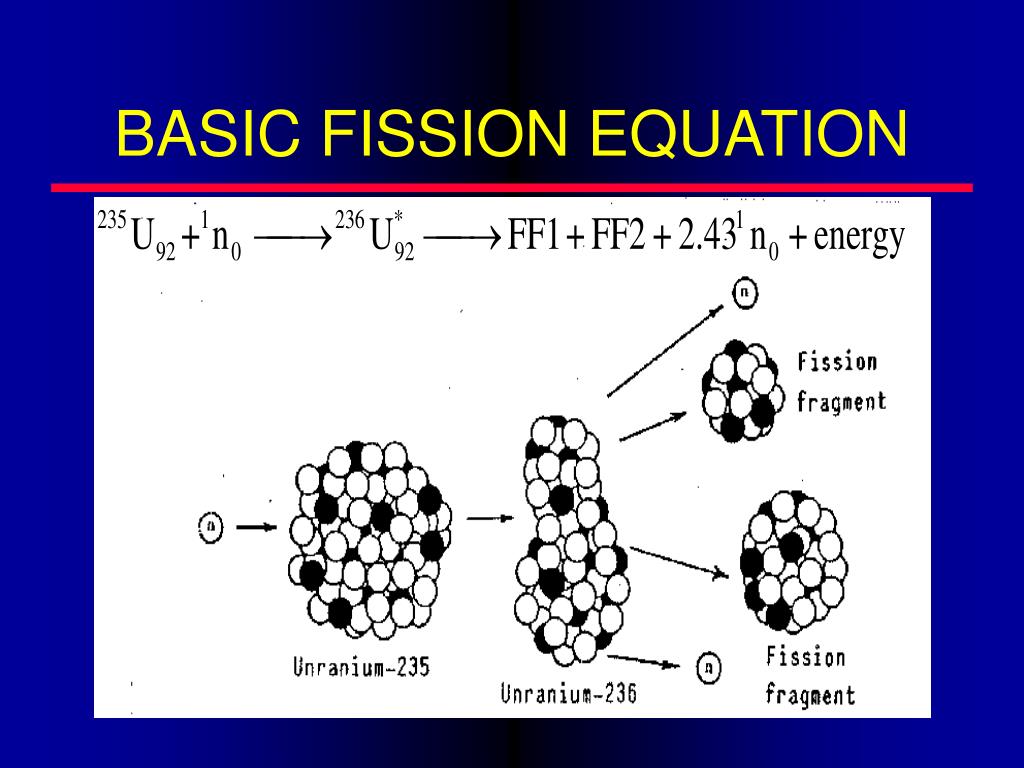
The term "nuclear reaction" may refer either to a change in a nuclide induced by collision with another particle or to a spontaneous change of a nuclide without collision. In principle, a reaction can involve more than two particles colliding, but because the probability of three or more nuclei to meet at the same time at the same place is much less than for two nuclei, such an event is exceptionally rare (see triple alpha process for an example very close to a three-body nuclear reaction). If a nucleus interacts with another nucleus or particle and they then separate without changing the nature of any nuclide, the process is simply referred to as a type of nuclear scattering, rather than a nuclear reaction. Thus, a nuclear reaction must cause a transformation of at least one nuclide to another. In nuclear physics and nuclear chemistry, a nuclear reaction is a process in which two nuclei, or a nucleus and an external subatomic particle, collide to produce one or more new nuclides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
